Melatonin more than for Sleep!
While melatonin works as a natural sleep regulator, it affects health in many other important ways as well.1 For example, melatonin:
| Is a potent antioxidant2 |
| Plays an important role in cancer prevention3 |
| Is important for brain, cardiovascular and gastrointestinal health4 |
| Boosts immune function in a variety of ways |
| May improve the treatment of certain bacterial diseases, including tuberculosis5 |
| Helps quell inflammation |
| May prevent or improve autoimmune diseases, including Type 1 diabetes6 |
| Is an important energy hormone7 — If your sleep efficiency is impaired, meaning you’re not sleeping as deeply as you should, for as long as is ideal, then your energy level is going to be adversely affected |
As noted in the Journal of Critical Care:8
“Melatonin is a versatile molecule, synthesized not only in the pineal gland, but also in many other organs. Melatonin plays an important physiologic role in sleep and circadian rhythm regulation, immunoregulation, antioxidant and mitochondrial-protective functions, reproductive control, and regulation of mood. Melatonin has also been reported as effective in combating various bacterial and v. infections.”
Melatonin — Research on Sepsis?
The Journal of Critical Care paper,9 published in 2010, further highlights the potential role of melatonin in the treatment of sepsis (blood poisoning), a life-threatening condition triggered by a systemic infection that causes your body to overreact and launch an excessive and highly damaging immune response.
Unless promptly diagnosed and treated, it can rapidly progress to multiple-organ failure and death. The cytokine storm response appears to be a primary way by which infections claims the lives of those who are immunocompromised and/or elderly. According to the Journal of Critical Care:10
“Melatonin is an effective anti-inflammatory agent in various animal models of inflammation and sepsis, and its anti-inflammatory action has been attributed to inhibition of nitric oxide synthase with consequent reduction of peroxynitrite formation, to the stimulation of various antioxidant enzymes thus contributing to enhance the antioxidant defense, and to protective effects on mitochondrial function and in preventing apoptosis.
In a number of animal models of septic shock, as well as in patients with septic disease, melatonin reportedly exerts beneficial effects to arrest cellular damage and multiorgan failure.”
In summary, melatonin appears to reverse septic shock symptoms by:11
- Decreasing synthesis of proinflammatory cytokines
- Preventing lipopolysaccharide (LPS)-induced oxidative damage, endotoxemia and metabolic alterations
- Suppressing gene expression of the bad form of nitric oxide, inducible nitric oxide synthase (iNOS)
- Preventing apoptosis (cell death)
According to the authors, the ways in which melatonin prevents septic shock are complex:
“Apart from action on the local sites of inflammation, melatonin also exerts its beneficial actions through a multifactorial pathway including its effects as immunomodulatory, antioxidant and antiapoptotic agent.”
FREE WEBINAR
Registar here.
Melatonin Ameliorates Cytokine Responses
In her email, Seneff goes on to cite a 2014 study13 in the Journal of Pineal Research which, like the Journal of Critical Care paper, points out that melatonin accumulates in mitochondria and has both antioxidant and anti-inflammatory activity that could be useful in the treatment of sepsis.
Melatonin is derived from serotonin, which in turn is derived from tryptophan, one of the three aromatic amino acids that are products of the shikimate pathway. Glyphosate famously disrupts the shikimate pathway in weeds, and this is believed to be the main mechanism by which it kills the weeds. Our gut bacteria also produce tryptophan via the shikimate pathway, so glyphosate can be expected to reduce the bioavailability of tryptophan as a precursor to melatonin.
The study mentioned above was a Phase 1 dose escalation study in healthy volunteers to evaluate the tolerability and health effects of melatonin at various dosages. They also assessed the effect of melatonin in an ex vivo whole blood model mimicking sepsis.
No adverse effects were reported for dosages ranging from 20 milligram (mg) to 100 mg, and the blood model testing revealed melatonin and its metabolite 6-hydroxymelatonin “had beneficial effects on sepsis-induced mitochondrial dysfunction, oxidative stress and cytokine responses …” The authors further explain:14
“Oxidative stress in patients with sepsis has been consistently described over the last 20 years. Mitochondrial dysfunction initiated by oxidative stress drives inflammation and is generally accepted as playing a major role in sepsis-induced organ failure.
It has been recognized that exogenous antioxidants may be useful in sepsis, and more recently, the potential for antioxidants acting specifically in mitochondria has been highlighted.
We showed previously that antioxidants targeted to mitochondria, including melatonin, reduced organ damage in a rat model of sepsis. Exogenous melatonin has potent antioxidant activity, and it accumulates throughout cells, particularly in mitochondria. Metabolites of melatonin also have antioxidant activity, and products from the reactions with oxidant species are also antioxidants.
In vitro models of sepsis show that melatonin and its major hydroxylated metabolite, 6-hydroxymelatonin, are both effective at reducing the levels of key inflammatory cytokines, oxidative stress, and mitochondrial dysfunction. In rat models of sepsis, melatonin reduces oxidative damage and organ dysfunction and also decreases mortality.
The dose needed for antioxidant action is thought to be considerably higher than that given for modulation of the sleep–wake cycle, but the actual dose required in man is unclear, particularly because the major bioactive effects of oral melatonin in the context of inflammation are likely to be mediated primarily by metabolite levels.”
Antiseptic Effects of Melatonin
More recently, a 2019 animal study15 in the journal Frontiers in Immunology discusses how melatonin can protect against polymicrobial sepsis — i.e., sepsis caused by more than one microbial organism — a hallmark of which is severe loss of lymphocytes through apoptosis, resulting in a twofold higher lethality than unimicrobial sepsis (sepsis caused by a single microbe).16
In this case, melatonin appears to offer protection by having an antibacterial effect on white blood cells called neutrophils. A high neutrophil count is an indicator for infection. According to the authors of the 2019 study:
“Melatonin treatment inhibited peripheral tissue inflammation and tissue damage … consequently reducing the mortality of the mice. We found that macrophages and neutrophils expressed melatonin receptors.
Upon depletion of neutrophils, melatonin-induced protection against polymicrobial infection failed in the mice, but melatonin treatment in macrophage-depleted mice attenuated the mice mortality resulting from polymicrobial sepsis.
Moreover, melatonin treatment promoted the development of the neutrophil extracellular trap (NET), which contributed to anti-bacterial activity during polymicrobial infection, whereas the phagocytic activities of neutrophils were inhibited by melatonin.
The data from this study support previously unexplained antiseptic effects of melatonin during a polymicrobial infection and could be potentially useful for human patients with sepsis.”
Understanding Infections
The potential role of melatonin in infection was also addressed in an extensive and fully referenced March 14, 2020, article by medical researcher Doris Loh, published in the Italian online magazine Evolutamente.17
One answer may be because the v. load in those with this infection is extremely high — 1,000 times higher than the v. load seen in SARS patients back in 2003.18
Preliminary research19 cited by Loh shows this infection the v. is actively replicating in the throat during the first five days after the onset of symptoms. This higher v. load could be what allows the v. to overwhelm even the immune system of some younger and healthier individuals.
Inflammasomes and Cytokine Storms
Another mechanism that can help explain the high virulence of this infection compared to SARS has to do with a furin cleavage site in the spike protein of the v. that wasn’t present in the SARS v.. As explained by Loh, “The presence of furins on almost all cell surfaces allow a dramatically increased ability to fuse to host cells, facilitating v. entry …” She adds:20
“Cleavage specificity can dictate the tropism and virulence of the virus. The fact that this infection has cleavage sites for furin enzymes renders this v. to be highly pathogenic, with the capacity to replicate in MULTIPLE tissues and organs due to how furins are utilized and distributed in the human body.
Furin-like cleavage in human coronaviruses have been associated with the development of neurological diseases where the invasiveness and efficient establishment of lower pathogenicity can result in persistent infection of the central nervous system.
Thus it was not a surprise when in early March of 2020, doctors from Beijing Ditan Hospital affiliated to Capital Medical University, a designated institution for this infection treatment, showed for the first time that this infection can attack the human central nervous system, causing symptoms of encephalitis.
The presence of furin enzymes on all cell surfaces cleaves and activates this infection in a wide range of tissues and organs. Activated infection then unleashes NLRP3 inflammasomes, initiating a flurry of immune reactions that can result in deadly cytokine storms."
As the name implies, inflammasomes are part of your natural immune response that senses the presence of pathogens and other factors that could pose a threat. When a pathogen is detected, inflammasomes are activated and start releasing proinflammatory cytokines.
The inflammasome NLRP3, specifically, has been identified as a key culprit in acute respiratory distress syndrome (ARDS) and acute lung injury, both of which are potential outcomes of this infection.21
The cytokine storm associated with this infection appears to be due to the virus’ ability to stimulate your immune response via viroporins, virally-encoded ion channel proteins that in turn trigger the activation of NLRP3 inflammasomes. As explained by Loh, the more functional a v. viroporins are, the greater its viral replication rate and subsequent pathogenesis.
Is Elevated Melatonin Sparing Young Children from this infection?
As noted by Loh, the fatality rate of infection increases linearly with age, with patients over the age of 80 having the highest death rate.22,23,24 As of yet, the explanation for why children are less likely to contract the infection or show symptoms even when they are infected,25 is unknown.
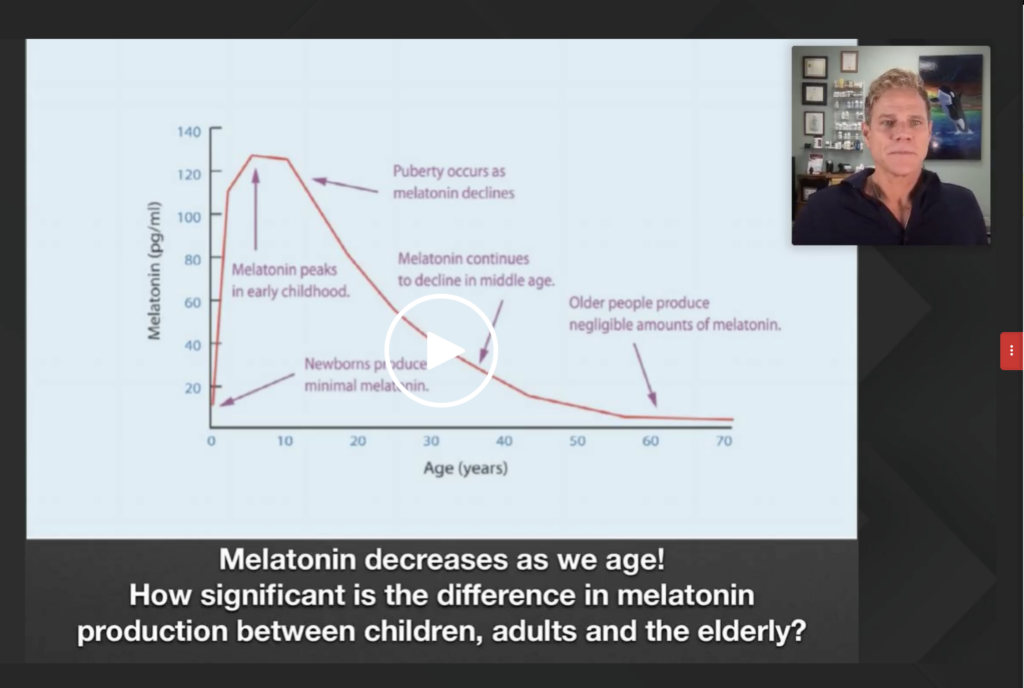
Loh, however, raises the possibility that it might have something to do with melatonin production. She cites research showing melatonin production peaks in early childhood, steadily dropping once puberty hits. By the time you’re in your late 50s, melatonin production drops to negligible levels.
“What does melatonin have to do with this infection?” Loh asks. The answer is that melatonin helps inhibit NLRP3 inflammasomes.
“The fact that the pro-inflammatory cytokine storm effects are induced by the activation of NLRP3 inflammasomes, the ability of melatonin to INHIBIT NLRP3 inflammasome elevates this powerful molecule to a truly unique position in the fight against this infection,” Loh writes.26
“This also means that if a patient, regardless of age, has adequate melatonin, the infectiousness will be greatly reduced, and the chances of developing ARDS/ALI significantly diminished.”
In her article, Loh references a number of scientific articles to support her claims. Aside from studies showing melatonin inhibits damage associated with sepsis, studies have also shown it can counteract severe inflammation and “markedly reduce pulmonary injury [and] lower infiltration of macrophages and neutrophils into lungs … by inhibiting the activation of NLRP3 inflammasomes,” thus protecting against acute lung injury.
Melatonin levels are also doubled in the third trimester of pregnancy compared to the first trimester,27,28 which may help explain why pregnant women infected with infections and their babies have gotten by fairly unscathed. A 2020 rodent study29 even demonstrated that melatonin can help protect against lung injury caused by mechanical ventilation intervention.
“The full therapeutic potential of melatonin in its ability to modulate the immune system, especially the critical function of suppressing cytokine storms to prevent progression of acute respiratory distress syndrome (ARDS) and respiratory failure in infected patients was clearly demonstrated in a study by Huang et al. (2019).
Huang et al. infected rodents with the highly lethal and infectious H1N1 influenza A virus. Co-treatment of these infected rodents with melatonin and an antiviral drug significantly increased their survival rates compared to mice treated only with antivirals alone,” Loh writes.
The role of NLRP3 inflammasomes in infection also adds support for the use of ascorbic acid (vitamin C), as ascorbic acid has been shown to inhibit NLRP3 Inflammasomes in a dose-dependent fashion by scavenging reactive oxygen species in the mitochondria.30,31
“Thus, the combined use of melatonin and ascorbic acid may prove to be most effective in the treatment for patients, especially those with cardiovascular and hypertension comorbidities,” Loh suggests.32
Avoid ACE Inhibitors During this infection Outbreak
Nutrients and drugs that might be best avoided during this outbreak are those that inhibit angiotensin-converting enzyme (ACE). What’s the problem with ACE inhibitors? As explained by Loh, ACE inhibitors increase expression of ACE2, and this infects host cells by binding to ACE2 receptors found on epithelial cells in your lungs, intestines, kidneys and blood vessels.33
ACE inhibitors, i.e., medications that relax and dilate your blood vessels, are commonly prescribed for high blood pressure, certain types of heart failure, stroke prevention and for the prevention and treatment of kidney disease. These drugs work by inhibiting the specific isoform of ACE, ACE1. As a consequence, ACE2 is upregulated. This is problematic with regard to this infection, because the v. gains entry to infect cells specifically via the ACE2 protein acting as a receptor.
Loh cites research34 published in The Lancet Respiratory Medicine suggesting that taking an ACE inhibitor, be it a medication or a nutritional supplement, may worsen your risk of v. infection and your risk of complications.
“Melatonin, nitric oxide and ascorbic acid (vitamin C) are all inextricably intertwined and deeply involved with ACE2,” Loh writes.35
“Melatonin, nitric oxide and ascorbic acid can reduce Viral virulence by inhibiting NLRP3 inflammasomes to stop the perpetuation of cytokine storms. Their critical roles in biochemical reactions and biological pathways that involve ACE2 must be fully explored as part of our fight against this infection.”
At the end of her article she offers suggestions for melatonin and vitamin C dosages, along with contraindications and timing of supplementation. I agree and studies show that higher doses of melatonin can safely be taken!
You should share this article with those you love and consider speaking to you heath care professional about a melatonin supplement. Of course this will not be a substitute for any medical care prescribed to you by your doctor.
FOR A DEEP DIVE INTO HOW MELATONIN WORKS ON IMMUNE SYSTEM AND HOW IT IS PART OF THE MATH PLUS. Ref
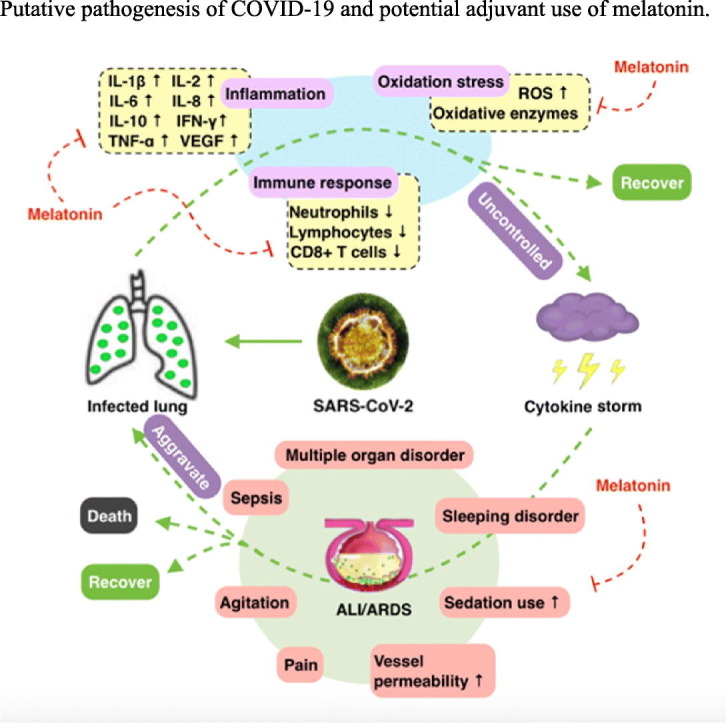
Melatonin as a potential adjuvant treatment
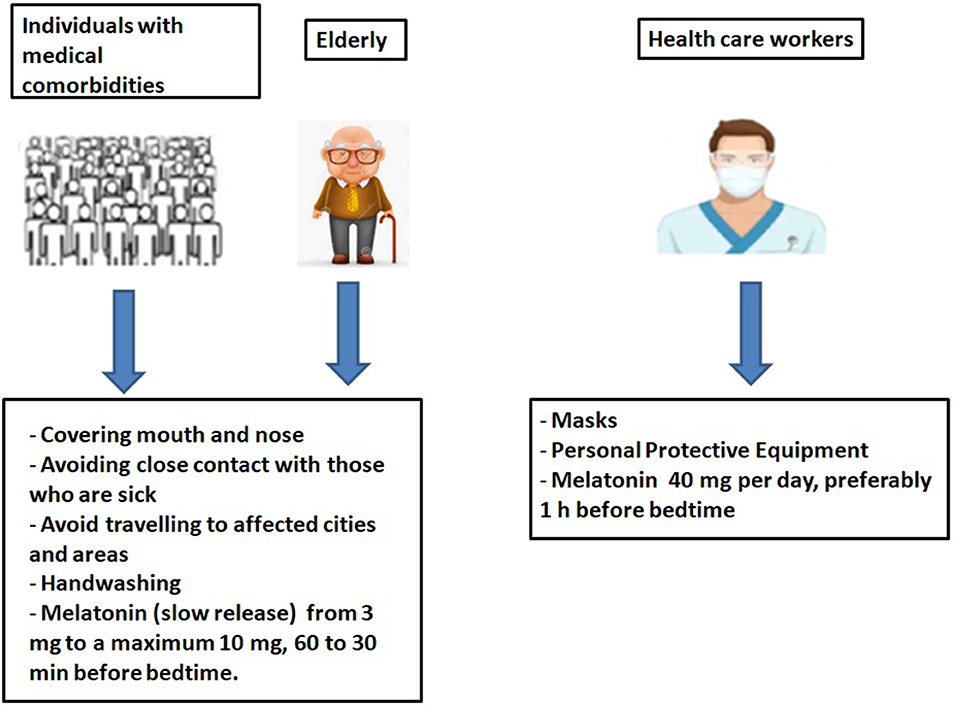
Dosages of melatonin. See the high doses they suggest in this article.
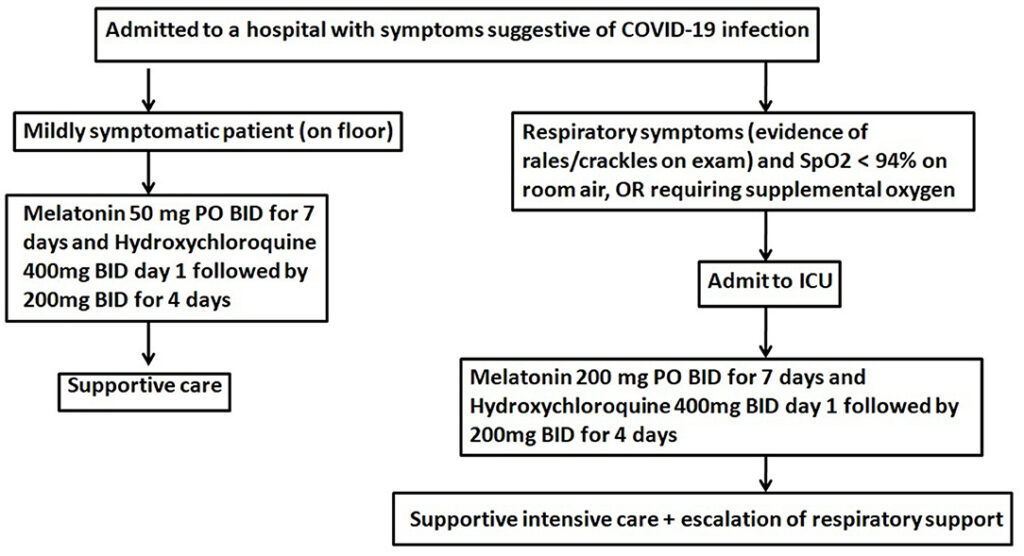
Algorithm for Use of Melatonin.